A) Addition of a substance to the left side of an equilibrium shifts it to the right.
B) Any stress to the right side of a chemical equilibrium results in a shift to the left to relieve the stress.
C) Heat added or taken away from a chemical equilibrium in which one side is exothermic will not shift the reaction.
D) Responses b and c are correct.
F) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
A heterogeneous catalyst will normally dissolve in the reaction mixture.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An elderly person comes to you on a winter day with blue fingers and toes,sluggish movement,and slurred speech.What condition do they most likely have?
A) hyperthermia
B) hypothermia
C) heat exhaustion
D) none of the choices
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement applies to a reaction at equilibrium?
A) The energy of activation has been supplied to bring the reaction to equilibrium.
B) The state of the equilibrium can be expressed as a ratio.
C) There are equal amounts of matter on either side of the reaction.
D) All of the responses are correct.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
A reaction rate can be described in terms of the change in concentration of either a reactant or a product.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following states of matter generally has the lowest entropy?
A) a crystalline solid
B) a liquid
C) a gas
D) two have the same entropy
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following products is not suitable to timed-release method for medications that are desired to have an extended effective level time?
A) appetite control materials
B) analgesics used for middle-aged people
C) fluoride compounds in a toothpaste
D) diuretics
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is assumed to be true in most explanations of how reactions occur?
A) reactant particles must collide with each other
B) catalysts must be present
C) energy must be absorbed as the reaction proceeds
D) more than one response is correct
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the reaction below,which of the following would take place once pure A and B are mixed together?
A + B  C
C
A) The concentration of C would increase for a time,then remain constant.
B) The concentration of A would increase for a time,then decrease.
C) The concentration of B would increase for a time,then remain constant.
D) More than one response is correct.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
Catalysts may lower the activation energy.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
For the reaction NO2 (g)+ N2O(g)  3 NO(g)the equilibrium constant expression is:
3 NO(g)the equilibrium constant expression is: 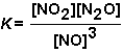
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In an exergonic process,the system _____ .
A) gains energy
B) loses energy
C) either gains or loses energy
D) no energy change at all
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about a K of 3.5 calculated for a specific chemical reaction at equilibrium is incorrect?
A) It was calculated by dividing the product of the products by that of the reactants.
B) It indicates that one side of the reaction has most of the matter present.
C) It is an evaluation of the extent of the two chemical reactions involved: the forward and the reverse reactions.
D) A K of 3.5 indicates that a catalyst will push the reaction to the right.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following will influence all reaction rates?
A) the presence of catalysts
B) the temperature of reactants
C) the concentration of reactants
D) more than one response is correct
F) All of the above
Correct Answer

verified
D
Correct Answer
verified
True/False
When a spontaneous process in accompanied by an energy increase,then a large entropy increase must also occur.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
CFC's such as CF2Cl2,are associated with the increase in chemical activity that degrades ozone.Which of the following relates to this phenomenon?
A) This is not serious because the only area of the planet involved is at the poles and there is very little life to be affected by the increase in UV.
B) The catalyzing of the ozone breakdown decreases the amount of UV at the surface of the planet.
C) The process by which the ozone is degraded is catalytic and can be controlled by the reduction of CFC's.
D) All of these responses are correct.
F) A) and C)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
What is the direct cause of a chemical reaction of the type E + F EF?
A) Catalysts are the direct cause of chemical reactions of this type.
B) The energy in the environment at room temperature causes the reaction.
C) The electromagnetic attraction between E and F draw them together.
D) Collisions occurring between E's and F's lead them to reaction.
F) B) and D)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
If we add a catalyst to the following equation,which way will the equilibrium shift?
CO + H2O + heat  CO2 + H2
CO2 + H2
A) to the left
B) will have no effect
C) to the right
D) not enough information.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following would be the equilibrium expression for the system given below?
N2(g) + 3F2(g)  2NF3(g)
2NF3(g)
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A process that gains or accepts energy as it takes place is a(n) _____ .
A) exergonic process
B) spontaneous process
C) endergonic process
D) all of these
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 87
Related Exams