Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the zeroth-order reaction: A → products,what will happen to the rate of reaction if the concentration of A is tripled?
A) The rate will be higher.
B) The rate will be tripled.
C) The rate will be one-third.
D) The rate will remain the same.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the first-order decomposition of A molecules (shaded spheres) in two vessels of equal volume.What is the half-life of decomposition in vessel (b) relative to the half-life of decomposition in vessel (a) ? 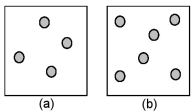
A) half-life in vessel (b) /half-life in vessel (a) = 4:1
B) half-life in vessel (b) /half-life in vessel (a) = 2:1
C) half-life in vessel (b) /half-life in vessel (a) = 3:2
D) half-life in vessel (b) /half-life in vessel (a) = 1:1
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a particular first-order reaction,it takes 48 minutes for the concentration of the reactant to decrease to 25% of its initial value.What is the value for rate constant (in s-1) for the reaction?
A) 1.0 × 10-4 s-1
B) 4.8 × 10-4 s-1
C) 6.0 × 10-3 s-1
D) 2.9 × 10-2 s-1
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The decomposition of dinitrogen pentoxide is described by the chemical equation 2 N2O5(g) → 4 NO2(g) + O2(g) If the rate of appearance of O2 is equal to 2.80 mol/min at a particular moment,what is the rate of disappearance of N2O5 at that moment?
A) 0.700 mol/min
B) 1.40 mol/min
C) 5.60 mol/min
D) 11.2 mol/min
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction that occurs in a Breathalyzer,a device used to determine the alcohol level in a person's bloodstream,is given below.If the rate of appearance of Cr2(SO4) 3 is 1.24 mol/min at a particular moment,what is the rate of disappearance of C2H6O at that moment? 2 K2Cr2O7 + 8 H2SO4 + 3 C2H6O → 2 Cr2(SO4) 3 + 2 K2SO4 + 11 H2O
A) 0.413 mol/min
B) 0.826 mol/min
C) 1.86 mol/min
D) 3.72 mol/min
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A mechanism for a naturally occurring reaction that destroys ozone is: Step 1: O3(g) + HO(g) → HO2(g) + O2(g) Step 2: HO2(g) + O(g) → HO(g) + O2(g) Which species is a catalyst?
A) HO
B) HO2
C) O
D) O3
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
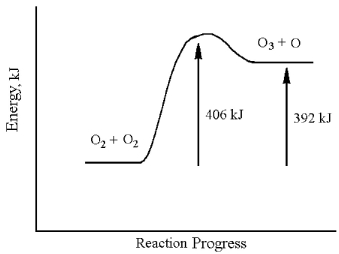 -What is the activation energy for the destruction of ozone?
-What is the activation energy for the destruction of ozone?
A) 14 kJ
B) 392 kJ
C) 406 kJ
D) none of these
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following reaction is first order in A and first order in B: A + B → Products Rate = k[A][B]
What is the rate constant k of this reaction in vessel (b) relative to the rate constant k of this reaction in vessel (a) ? Each vessel has the same volume.Shaded spheres represent A molecules,and unshaded spheres represent B molecules. ![The following reaction is first order in A and first order in B: A + B → Products Rate = k[A][B] What is the rate constant k of this reaction in vessel (b) relative to the rate constant k of this reaction in vessel (a) ? Each vessel has the same volume.Shaded spheres represent A molecules,and unshaded spheres represent B molecules. A) rate constant k in vessel (b) /rate constant k in vessel (a) = 1:2 B) rate constant k in vessel (b) /rate constant k in vessel (a) = 1:1 C) rate constant k in vessel (b) /rate constant k in vessel (a) = 2:1 D) rate constant k in vessel (b) /rate constant k in vessel (a) = 4:1](https://d2lvgg3v3hfg70.cloudfront.net/TB4940/11ea7e2d_d159_e924_a2f7_0134c79a9143_TB4940_00.jpg)
A) rate constant k in vessel (b) /rate constant k in vessel (a) = 1:2
B) rate constant k in vessel (b) /rate constant k in vessel (a) = 1:1
C) rate constant k in vessel (b) /rate constant k in vessel (a) = 2:1
D) rate constant k in vessel (b) /rate constant k in vessel (a) = 4:1
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
 -What is the activation energy for the formation of ozone?
-What is the activation energy for the formation of ozone?
A) 14 kJ
B) 392 kJ
C) 406 kJ
D) none of these
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the first-order reaction A → B in which A molecules (shaded spheres) are converted to B molecules (unshaded spheres) . 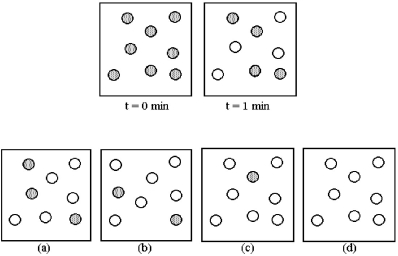 -Which drawing (a) -(d) represents the reaction mixture at t = 3 minutes?
-Which drawing (a) -(d) represents the reaction mixture at t = 3 minutes?
A) drawing (a)
B) drawing (b)
C) drawing (c)
D) drawing (d)
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The decomposition of dinitrogen pentoxide is described by the chemical equation 2 N2O5(g) → 4 NO2(g) + O2(g) If the rate of appearance of NO2 is equal to 0.560 mol/min at a particular moment,what is the rate of appearance of O2 at that moment?
A) 0.140 mol/min
B) 0.280 mol/min
C) 1.12 mol/min
D) 2.24 mol/min
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hydrogen peroxide decomposes to water and oxygen according to the reaction below: 2 H2O2(aq) → 2 H2O(l) + O2(g)
In the presence of large excesses of I- ion,the following set of data is obtained.What is the average rate of disappearance of H2O2(aq) in M/s in the first 45.0 seconds of the reaction if 1.00 L of H2O2 reacts at 25°C and 1.00 atm pressure? 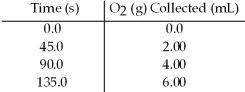
A) 9.09 × 10-7 M/s
B) 3.63 × 10-6 M/s
C) 4.33 × 10-5 M/s
D) 1.64 × 10-4 M/s
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A concentration-time study of the gas phase reaction 2 A3 → 3 A2 produced the data in the table below. Time (s) [A3] (M) [A2] (M) 0 4.00 × 10-4 0 10 2.00 × 10-4 3.00 × 10-4 20 1.00 × 10-4 4.50 × 10-4 30 5.00 × 10-5 ? What is the concentration of A2 after 30 seconds?
A) 5.00 × 10-4 M
B) 5.25 × 10-4 M
C) 5.50 × 10-4 M
D) 6.00 × 10-4 M
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the overall reaction order for the reaction that has the rate law: Rate = k[Cl ]2[N2]?
A) zero order
B) first order
C) second order
D) third order
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider a reaction that occurs by the following one-step mechanism:
A2 + B2 → 2 AB
The potential energy profile for this reaction is shown below. 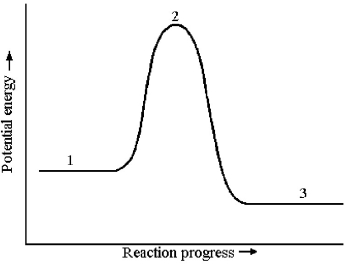 -The energy of reaction,ΔE,is given by the difference in energy between which two reaction stages?
-The energy of reaction,ΔE,is given by the difference in energy between which two reaction stages?
A) reaction stage 2 - reaction stage 1
B) reaction stage 2 - reaction stage 3
C) reaction stage 1 - reaction stage 3
D) reaction stage 3 - reaction stage 1
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following reaction is second order in A and first order in B: A + B → Products Rate = k[A]2[B]
What is the initial rate of this reaction in vessel (b) relative to the initial rate of this reaction in vessel (a) ? Each vessel has the same volume.Shaded spheres represent A molecules,and unshaded spheres represent B molecules present at the beginning of the reaction. ![The following reaction is second order in A and first order in B: A + B → Products Rate = k[A]<sup>2</sup>[B] What is the initial rate of this reaction in vessel (b) relative to the initial rate of this reaction in vessel (a) ? Each vessel has the same volume.Shaded spheres represent A molecules,and unshaded spheres represent B molecules present at the beginning of the reaction. A) rate in vessel (b) /rate in vessel (a) = 1:2 B) rate in vessel (b) /rate in vessel (a) = 1:1 C) rate in vessel (b) /rate in vessel (a) = 2:1 D) rate in vessel (b) /rate in vessel (a) = 4:1](https://d2lvgg3v3hfg70.cloudfront.net/TB4940/11ea7e2d_d159_4ce1_a2f7_a3236f892a95_TB4940_00.jpg)
A) rate in vessel (b) /rate in vessel (a) = 1:2
B) rate in vessel (b) /rate in vessel (a) = 1:1
C) rate in vessel (b) /rate in vessel (a) = 2:1
D) rate in vessel (b) /rate in vessel (a) = 4:1
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
For the reaction shown below,if the rate of formation of O2 is 6.5 × 10-6 M/s,the rate of decomposition of N2O5 over the same time interval is ________. 2 N2O5(g)→ 4 NO2(g)+ O2(g)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Chlorine reacts with chloroform according to the reaction given below: Cl2 + CHCl3 → CCl4 + HCl When the initial concentration of Cl2 is doubled the reaction rate increases by a factor of 1.41.What is the order of the reaction with respect to Cl2?
A) -1/2
B) -1
C) 1/2
D) 2
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following pictures represent the progress of a reaction in which two A molecules combine to give a more complex molecule A2,2A → A2. 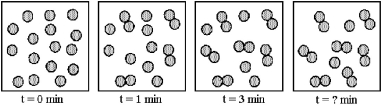 -What is the order of reaction with respect to A?
-What is the order of reaction with respect to A?
A) 0
B) 1/2
C) 1
D) 2
F) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 190
Related Exams