A) increasing the concentration of reactant A
B) increasing the concentration of reactant B
C) increasing the temperature of the reaction
D) adding a catalyst for the reaction
E) All of these will increase the rate.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Nitrogen dioxide decomposes to nitric oxide and oxygen via the reaction: 2NO2 (g) → 2NO (g) + O2 (g) In a particular experiment at 300 °C, [NO2] drops from 0.0100 to 0.00800 M in 100 s. The rate of appearance of O2 for this period is ________ M/s.
A) 1.0 × 10-5
B) 2.0 × 10-5
C) 4.0 × 10-5
D) 2.0 × 10-3
E) 4.0 × 10-3
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Of the following, only ________ is a valid unit for reaction rate.
A) M/s
B) mmol/mL
C) mol/g
D) g/L
E) atm/g
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A catalyst can increase the rate of a reaction ________.
A) by changing the value of the frequency factor (A)
B) by increasing the overall activation energy (Ea) of the reaction
C) by lowering the activation energy of the reverse reaction
D) by providing an alternative pathway with a lower activation energy
E) All of these are ways that a catalyst might act to increase the rate of reaction.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A flask is charged with 0.124 mol of A and allowed to react to form B according to the reaction A(g) →B(g) . The following data are obtained for [A] as the reaction proceeds: ![A flask is charged with 0.124 mol of A and allowed to react to form B according to the reaction A(g) →B(g) . The following data are obtained for [A] as the reaction proceeds: -The average rate of disappearance of A between 20 s and 40 s is ________ mol/s. A) 8.5 × 10<sup>-4</sup> B) 1.7 × 10<sup>-3</sup> C) 590 D) 7.1 × 10<sup>-3</sup> E) 1.4 × 10<sup>-3</sup>](https://d2lvgg3v3hfg70.cloudfront.net/TB2701/11ea7cce_1f63_2626_a2ab_9b6fd935b612_TB2701_00_TB2701_00_TB2701_00_TB2701_00_TB2701_00_TB2701_00.jpg) -The average rate of disappearance of A between 20 s and 40 s is ________ mol/s.
-The average rate of disappearance of A between 20 s and 40 s is ________ mol/s.
A) 8.5 × 10-4
B) 1.7 × 10-3
C) 590
D) 7.1 × 10-3
E) 1.4 × 10-3
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction A → B is first order in [A]. Consider the following data. ![The reaction A → B is first order in [A]. Consider the following data. The rate constant for this reaction is ________ s<sup>-1</sup>. A) 0.014 B) 0.030 C) 0.013 D) 3.0 E) 3.1 × 10<sup>-3</sup>](https://d2lvgg3v3hfg70.cloudfront.net/TB2701/11ea7cce_1f64_acce_a2ab_fb26179093f9_TB2701_00.jpg) The rate constant for this reaction is ________ s-1.
The rate constant for this reaction is ________ s-1.
A) 0.014
B) 0.030
C) 0.013
D) 3.0
E) 3.1 × 10-3
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The concentration of S2O82- remaining at 1600 s is ________ M.
A) 0.036
B) 0.014
C) 0.043
D) 0.064
E) 0.029
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The initial concentration of reactant in a first-order reaction is 0.27 M. The rate constant for the reaction is 0.75 s-1. What is the concentration (mol/L) of reactant after 1.8 s?
A) 6.0
B) 1.7
C) 0.070
D) 0.012
E) 1.0
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
Reaction rates are affected by reactant concentrations and temperature. This is accounted for by the ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The rate of disappearance of HBr in the gas phase reaction 2HBr (g) → H2 (g) + Br2 (g) Is 0.190 M s-1 at 150 °C. The rate of appearance of Br2 is ________ M s-1.
A) 2.63
B) 0.095
C) 0.0361
D) 0.380
E) 0.436
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the elementary reaction NO3 + CO → NO2 + CO2
The molecularity of the reaction is ________, and the rate law is ![For the elementary reaction NO<sub>3</sub> + CO → NO<sub>2</sub> + CO<sub>2</sub> The molecularity of the reaction is ________, and the rate law is A) 2, k[NO<sub>3</sub>][CO] B) 4, k[NO<sub>3</sub>][CO][NO<sub>2</sub>][CO<sub>2</sub>] C) 2, k[NO<sub>2</sub>][CO<sub>2</sub>] D) 2, k[NO<sub>3</sub>][CO]/[NO<sub>2</sub>][CO<sub>2</sub>] E) 4, k[NO<sub>2</sub>][CO<sub>2</sub>]/[NO<sub>3</sub>][CO]](https://d2lvgg3v3hfg70.cloudfront.net/TB2701/11ea7cce_1f62_b0f3_a2ab_1d7683edb257_TB2701_11.jpg)
A) 2, k[NO3][CO]
B) 4, k[NO3][CO][NO2][CO2]
C) 2, k[NO2][CO2]
D) 2, k[NO3][CO]/[NO2][CO2]
E) 4, k[NO2][CO2]/[NO3][CO]
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The concentration of S2O82- remaining at 800 s is ________ M.
A) 0.046
B) 0.076
C) 4.00 × 10-3
D) 0.015
E) 0.041
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At elevated temperatures, nitrogen dioxide decomposes to nitrogen oxide and oxygen: NO2 (g) → NO (g) + ![At elevated temperatures, nitrogen dioxide decomposes to nitrogen oxide and oxygen: NO<sub>2 </sub>(g) → NO (g) + O<sub>2 </sub>(g) The reaction is second order in NO<sub>2</sub> with a rate constant of 0.543 M<sup>-1</sup>s<sup>-1</sup>at 300 °C. If the initial [NO<sub>2</sub>] is 0.260 M, it will take ________ s for the concentration to drop to 0.075 M. A) 3.34 B) 0.0880 C) 2.29 D) 0.299 E) 17.5](https://d2lvgg3v3hfg70.cloudfront.net/TB2701/11ea7cce_1f65_4910_a2ab_c59f7627143b_TB2701_11.jpg) O2 (g)
The reaction is second order in NO2 with a rate constant of 0.543 M-1s-1at 300 °C. If the initial [NO2] is 0.260 M, it will take ________ s for the concentration to drop to 0.075 M.
O2 (g)
The reaction is second order in NO2 with a rate constant of 0.543 M-1s-1at 300 °C. If the initial [NO2] is 0.260 M, it will take ________ s for the concentration to drop to 0.075 M.
A) 3.34
B) 0.0880
C) 2.29
D) 0.299
E) 17.5
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The kinetics of the reaction below were studied and it was determined that the reaction rate did not change when the concentration of B was tripled. The reaction is ________ order in B. A + B → P
A) zero
B) first
C) second
D) third
E) one-half
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The data in the table below were obtained for the reaction:
2 ClO2 (aq) + 2 OH- (aq) → ClO3- (aq) + ClO2- (aq) + H2O (1) 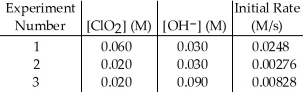 -What is the magnitude of the rate constant for the reaction?
-What is the magnitude of the rate constant for the reaction?
A) 1.15 × 104
B) 4.6
C) 230
D) 115
E) 713
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A reaction was found to be second order in carbon monoxide concentration. The rate of the reaction ________ if the [CO] is doubled, with everything else kept the same.
A) doubles
B) remains unchanged
C) triples
D) increases by a factor of 4
E) is reduced by a factor of 2
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A flask is charged with 0.124 mol of A and allowed to react to form B according to the reaction A(g) →B(g) . The following data are obtained for [A] as the reaction proceeds: ![A flask is charged with 0.124 mol of A and allowed to react to form B according to the reaction A(g) →B(g) . The following data are obtained for [A] as the reaction proceeds: -The average rate disappearance of A between 20 s and 30 s is ________ mol/s. A) 5.0 × 10<sup>-4</sup> B) 1.6 × 10<sup>-2</sup> C) 1.5 × 10<sup>-3</sup> D) 670 E) 0.15](https://d2lvgg3v3hfg70.cloudfront.net/TB2701/11ea7cce_1f63_2626_a2ab_9b6fd935b612_TB2701_00_TB2701_00_TB2701_00_TB2701_00_TB2701_00_TB2701_00.jpg) -The average rate disappearance of A between 20 s and 30 s is ________ mol/s.
-The average rate disappearance of A between 20 s and 30 s is ________ mol/s.
A) 5.0 × 10-4
B) 1.6 × 10-2
C) 1.5 × 10-3
D) 670
E) 0.15
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction 2NO2 → 2NO + O2 Follows second-order kinetics. At 300 °C, [NO2] drops from 0.0100 M to 0.00650 M in 100.0 s. The rate constant for the reaction is ________ M-1s-1.
A) 0.096
B) 0.65
C) 0.81
D) 1.2
E) 0.54
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The half-life for a first order rate law depends on the starting concentration.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The overall order of a reaction is 1. The units of the rate constant for the reaction are ________.
A) M/s
B) M-1s-1
C) 1/s
D) 1/M
E) s/M2
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 134
Related Exams