A) the half-life of a radioisotope.
B) the energy deposited per gram of an object.
C) the biological damage produced.
D) the accumulation of fission products.
E) the number of ions per centimeter.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rubidium-87 decays by beta decay with a half-life of 4.9 * 1010 yr. How many 87Rb atoms are in a moon rock sample that has a rubidium decay rate of 3,500 disintegrations per hour?
A) 9.0 * 1016 atoms
B) 4.3 * 10-4 atoms
C) 2.2 * 1018 atoms
D) 2.5 * 1014 atoms
E) 1.7 * 1014 atoms
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Protactinium-234 has a half-life of 1 minute. How much of a 400. g sample protactinium would remain after 1 minute?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sulfur-35 decays by beta emission. The decay product is
A) ![]() .
.
B) ![]() .
.
C) ![]() .
.
D) ![]() .
.
E) ![]() .
.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Calculate the binding energy per nucleon of an Al-27 nucleus given the following masses: 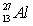 : 26.981541 amu;
: 26.981541 amu; 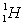 : 1.007838 amu;
: 1.007838 amu; 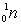 : 1.008665 amu.
: 1.008665 amu.
Correct Answer

verified
1.34 * 10<...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
When atoms of beryllium-9 are bombarded with alpha particles, neutrons are produced. What new isotope is also formed? 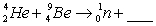
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many 14C atoms are in a charcoal sample that has a decay rate of 3,500 disintegrations per min? (For 14C, t1/2 = 5,730 yr.)
A) 2.9 * 107 atoms
B) 8.0 * 10-7 atoms
C) 1.4 * 1014 atoms
D) 1.5 * 1013 atoms
E) 6.02 * 1020 atoms
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the following reaction, identify X. 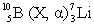
A) ( )
B) n
C) p
D) ![]()
E) ( )
G) C) and E)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
The energy released by the sun is the result of
A) natural radioactivity.
B) nuclear fusion.
C) combustion of hydrogen.
D) photosynthesis.
E) nuclear fission.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Polonium-208 is an alpha emitter with a half-life of 2.90 years. How many milligrams of polonium from an original sample of 2.00 mg will remain after 8.00 years?
A) 0.147 mg
B) 0.296 mg
C) 0.725 mg
D) 6.77 mg
E) 1.90 mg
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The only stable isotope of iodine is iodine-127. Predict the mode of decay of  .
.
A) alpha emission
B) beta emission
C) positron emission
D) electron capture
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Decay of lutetium-167 by electron capture yields
A) ytterbium-167.
B) lutetium-166.
C) thulium-163.
D) tantalum-171.
E) hafnium-167.
G) A) and C)
Correct Answer

verified
A
Correct Answer
verified
True/False
A plot of the number of neutrons versus the number of protons in various isotopes produces a "belt of stability." Isotopes below the belt of stability (i.e., with a smaller neutron-to-proton ratio)decay by beta particle emission.
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
Predict which nucleus is less stable, 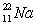 or
or 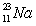 .
.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Alpha particles are identical to
A) protons.
B) helium atoms.
C) hydrogen atoms.
D) helium nuclei.
E) electrons.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Essay
Balance the equation 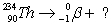 .
.
Correct Answer

verified
Correct Answer
verified
Short Answer
Strontium-90 has a half-life of 28.8 years. How much strontium-90 was present initially, if after 144 years 10.0 g remain?
Correct Answer

verified
Correct Answer
verified
Essay
Balance the equation 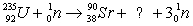 .
.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The half-life of 90Sr is 29 years. What fraction of the atoms in a sample of 90Sr would remain 175 years later?
A) 0.17
B) 0.12
C) 0.062
D) 0.015
E) 0.50
G) A) and B)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Gamma-rays cause radiation damage when they interact with matter by producing
A) ions and free radicals.
B) isotopes.
C) daughter products.
D) oxidation.
E) reduction.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 112
Related Exams